
For years, noted chemist and synthetic life researcher Steven Benner has talked about the necessary role of the element boron in the origin of life.
Without boron, he has found, many of the building blocks needed to form the earliest self-replicating ribonucleic acid (RNA) fall apart when they come into contact with water, which is nonetheless needed for the chemistry to succeed. Only in the presence of boron, Benner found and has long argued, can the formation of RNA and later DNA proceed.
Now, to the delight of Benner and many other scientists, the Curiosity team has found boron on Mars. In fact, as Curiosity climbs the mountain at the center of Gale Crater, the presence of boron has become increasingly pronounced.
And to make the discovery all the more meaningful to Benner, the boron is being found in rock veins. So it clearly was carried by water into the fractures, and was deposited there some 3.5 billion years ago.
Combined with earlier detections of phosphates, magnesium, peridots, carbon and other essential elements in Gale Crater, Benner told me, “we have found on Mars an environment entirely consistent with a what we consider conducive for the origin of life.
“Is it likely that life arose? I’d say yes…perhaps even, hell yes. But it’s also true that an environment conducive to the formation of life isn’t necessarily one conducive to the long-term survival of life.”

Another factor in the Mars-as-habitable story from Benner’s view is that there has never been the kind of water world there that many believe existed on early Earth.
While satellites orbiting Mars and now Curiosity have made it abundantly clear that early Mars also had substantial water in the form of lakes, rivers, streams and perhaps an localized ocean, it was clearly never covered in water.
And that’s good for the origin of life, Benner said.

“We think that a largely arid environment, with water present but not everywhere, is the best one for life to begin. Mars had that but Earth, well, maybe not so much. The problem is how to concentrate the makings of RNA, of life, in a vast ocean. It’s like making a cake in water — all the ingredients will float away.
“But the mineral ensemble they’ve discovered and given us is everything we could have asked for, and it was on a largely dry Mars,” he said. “So they’ve kicked the ball back to us. Now we have to go back to our labs to enrich the chemistry around this ensemble of minerals.”
In his labs, Benner has already put together a process — he calls it his discontinuous synthesis model — whereby all the many steps needed to create RNA and therefore life have been demonstrated to be entirely possible.
What’s missing is a continuous model that would show that process at work, starting with a particular atmosphere and particular minerals and ending up with RNA. That’s something that requires a lot more space and time than any lab experiments would provide.
“This is potentially what Mars provides,” he said,
Benner, it should be said, is not a member of the Curiosity team and doesn’t speak for them.
But his championing of boron as a potentially key element for the origin of life was noted as a guide by one of the Curiosity researchers during a press conference with team members at the American Geophysical Union Dec. 13 in San Francisco. It was at that gathering that the detection of the first boron on Mars was announced.
Benner said he has been in close touch with the two Curiosity instrument teams involved in the boron research and was most pleased that his own boron work — and that of at least one other researcher — had helped inspire the search for and detection of the element on Mars. That other researcher, evolutionary biologist James Stephenson, had detected boron in a meteorite from Mars.
Patrick Gasda, a postdoctoral researcher at Los Alamos National Laboratory, is a member of the Chemistry and Camera (ChemCam) instrument team which identified the boron at Gale Crater. The instrument uses laser technology to identify chemical elements in Martian rocks.
Gasda said at AGU that if the boron they found in calcium sulfate rock veins on Mars behaves there as it does on Earth, then the environment was conducive to life. The ancient groundwater that formed these veins would have had temperatures in the 0-60 degrees Celsius (32-140 degrees Fahrenheit) range, he said, with a neutral-to-alkaline pH.
While the presence of boron (most likely the mineral form borate, Benner said) has increased as the rover has climbed Mount Sharp, the element still makes up only one-tenth of one percent of the rock composition. But to stabilize that process of making RNA, that’s enough.
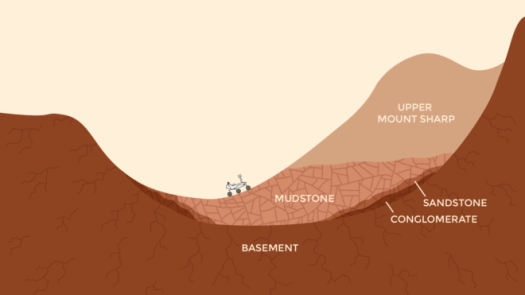
Benner’s view of Gale Crater and Mars as entirely habitable is not new — the Curiosity team has been saying roughly the same for several years now. But with four full years on Mars the rover keeps adding to the habitability story, and that was the central message from Curiosity scientists speaking at the AGU press conference.
As the rover examines higher, younger layers, the researchers said they were especially impressed by the complexity of the ancient lake environments at Gale when sediments were being deposited, and also the complexity of the groundwater interactions after the sediments were buried.
“There is so much variability in the composition at different elevations, we’ve hit a jackpot,” said John Grotzinger of Caltech, and formerly the mission scientist for Curiosity.
“A sedimentary basin such as this is a chemical reactor. Elements get rearranged. New minerals form and old ones dissolve. Electrons get redistributed. On Earth, these reactions support life.”
This kind of reactivity occurs on a gradient based on the strength of a chemical at donating or receiving electrons. Transfer of electrons due to this gradient can provide energy for life.

While habitability is key to Curiosity’s mission on Mars, much additional science is being done that has different goals or looks more indirectly at the planet’s ancient (or possibly current) ability to support life. Understanding the ancient environmental history of Gale Crater and Mars is a good example.
For instance, the Curiosity team is now undertaking a drilling campaign in progressively younger rock layers, digging into four sites each spaced about 80 feet (about 25 meters) further uphill. Changes in which minerals are present and in what percentages they exist give insights into some of that ancient history.
One clue to changing ancient conditions is the presence of the mineral hematite, a form of the omnipresent iron oxide on Mars. Hematite has replaced magnetite as the dominant iron oxide in rocks Curiosity has drilled recently, compared with the site where Curiosity first found lake bed sediments.
Thomas Bristow of NASA Ames Research Center, who works with the Chemistry and Mineralogy (CheMin) laboratory instrument inside the rover, said Mars is sending a signal. Both forms of iron oxide (hematite and magnetite) were deposited in mudstone in what was once the bottom of a lake, but the increased abundance of hematite higher up Mount Sharp suggests conditions were warmer when it was laid down. There also was probably more interaction between the atmosphere and the sediments.
On a more technical level, an increase in hematite relative to magnetite also indicates an environmental change towards a stronger tug on the iron oxide electrons, causing a greater degree of oxidation (the loss of electrons) in the iron. That would likely be caused by changing atmospheric conditions.
It’s all part of putting together the jigsaw puzzle of Mars circa 3.5 billion years ago.

Returning to the boron story, Benner said that the discovered presence of all the chemicals his group believes are necessary to ever-so-slowly move from prebiotic chemistry to biology provides an enormous opportunity. Because of plate tectonics on Earth and the omnipresence of biology, the conditions and environments present on early Earth when life first arose were long ago destroyed.
But on Mars, the apparent absence of those most powerful agents of change means it’s possible to detect, observe and study conditions in a changed but intact world that just might have given rise to life on Mars. Taken a step further, Mars today could provide new and important insights into how life arose on Earth.
And then there’s the logic of what finding signs of ancient, or perhaps deep-down surviving life on Mars would mean to the larger search for life in the cosmos.
That life exists on one planet among the hundreds of billions we now know are out there suggests that other planets — which we know have many or most of the same basic chemicals as Earth — might have given rise to life as well.
And if two planets in one of those many, many solar system have produced and supported life, then the odds go up dramatically regarding life on other planets.
One planet with life could be an anomaly. Two nearby planets with life, even if its similar, are a trend.
