We live in a dangerous universe. We know about meteor and comets, about harmful radiation that could extinguish life without an electromagnetic shield, about major changes in climate that are both natural and man-made.
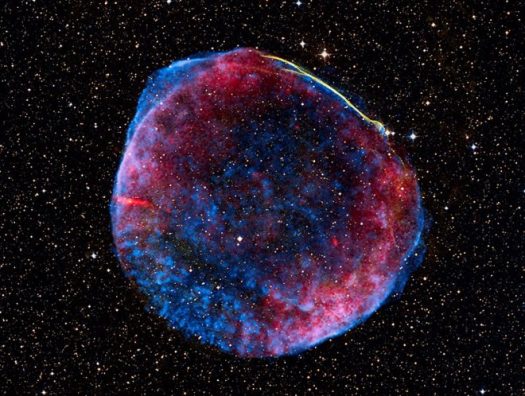
There’s another risk out there that some scientists assert could cause large-scale extinctions even though it would occur scores of light-years away. These are supernovae – explosions of massive stars that both create and spread the heavy elements needed for life and send out high energy cosmic rays that can travel far and cause enormous damage.
As with most of these potential threats, they fortunately occur on geological or astronomical time scales rather than human ones. But that doesn’t mean they don’t happen.
At the recent Astrobiology Science Conference (AbSciCon) a series of talks focused on that last threat – starting with a talk on “When Stars Attack.”
And together five different presenters made a persuasive case that Earth was on the receiving end of a distant supernova explosion some two to three million years ago, and probably around 7 or 8 million years ago as well. The effects of the cosmic ray bombardment have been debated and disputed, but the evidence for the occurrences is based on the rock record and is now strong.
“The evidence is there on the ocean floor: in rocky crusts, nodules and sediment,” said Brian Fields, professor of astronomy at University of Illinois. “We’ve been able to date it and provide some idea of how far away the star blew up.” The answer is between about 90 and 300 light-years.

“Supernova explosions happen all the time– on average every 30 years in our galaxy, though they are most often distant and obscured from view,” Fields said. “They generate cosmic rays that can spread through the galaxy for 30 million years. These are the cosmic rays that make carbon-14 and can threaten astronauts in space. But that’s not what we’re focused on — we look at the ones that are close to us and could have a far more dramatic effect, and they are pretty rare.”
What is deemed to be the “kill zone” for a planet nearby a supernova is 30 light-years; the high energy particles from an explosion that close would, he said, likely end all or most life on Earth by setting into motion a variety of atmospheric and surface changes. Fields there is no evidence of such a close and damaging supernove within the past 10 million years, the period that has been studied with some rigor.
But because a close supernova explosion hasn’t happened recently doesn’t mean that it didn’t happened during earlier times. Or that it couldn’t happen in the far future.
“By nailing the signal of a close but not ‘kill zone’ supernova two to three million years ago, and most likely another at 7 to 8 million years ago, we make the case that supernova can and do have significant effects on Earth.”
The community of scientists who study supernovae and their effects on Earth, both potential and known, is small, and has been most active in the past decade. There was an earlier time when scientists focused on supernovae as the potential cause for the massive dinosaur extinction, but the field shrank with confirmation in 1990 that a six-mile wide meteor landed on Mexico’s Yucatan Peninsula about 65 million years ago and was the likely cause of the global extinction.

But now, with the advent of new theories and some very high tech and precise measuring the field and subject has come to life, with research nodes in Germany, Australia and the American Midwest.
The key to understanding the effects of distant supernovae on Earth involves a radioactive isotope of iron, iron-60. It’s one of the many elements known to be sent into the cosmos by the massive thermonuclear blasts that define a supernova, that send out shock waves capable of spurring the formation of new stars as well as providing the universe with the heavier chemical elements needed to form everything from planets to genes.
It was the young Fields and colleagues who theorized some two decades ago that iron-60 could be a telltale sign of a relatively nearby supernova. He told me that no other significant sources of iron-60 are known to exist, and so if it were found on Earth scientists would know where it came from.
With a half-life of some three million years, the iron-60 would be a potentially strong signal for that length of time and and then a weaker but potentially detectable signal after that.
The question was how do you find iron-60 on Earth? The answer came from the bottom of the ocean.
First in 1999 a group from the Technical University of Munich in Germany identified some iron-60 in iron-manganese crustal rocks at the bottom of the Pacific, and then in 2013 reported finding the telltale isotope in not only rocks but also in nodules and most important in fossil bacteria and sea-floor sediments. They used ultra-sensitive accelerator mass spectrometry to isolate and identify the iron-60, which they reported was deposited some 1.6 to 3 million years ago.

Last year as well the Australian group, led by Anton Wallner of the Australian National University, found the iron-60 to be deposited globally and to have arrived within the same general time frame. And Gunther Korschinek, a physicist at the Technical University of Munich involved in the initial German iron-60 detections, led a team that found elevated amounts of iron-60 in lunar soil samples brought from to moon back to Earth during the Apollo program.
As Fields put it, the studies together gave a clear signal of a supernova explosion, or series of explosions, at 2 to 3 million years ago, and a less clear but likely signal of the same at 7 to 8 million years ago.
Since Fields and other scientists were presenting during the AbSciCon conference, the talks not surprisingly focused on potential biological implications of supernova explosions. And while supernova impacts on the biosphere are not particularly well understood, a number of intriguing theories were presented.
Brian Thomas of Washburn University described how cosmic rays from close supernova would significantly increase levels of electrically charged elements and molecules in the atmosphere, lasting thousands of years. In the upper atmosphere this would have the effect of setting into motion a chemical cascade that would deplete stratospheric ozone. In the lower atmosphere, the effect would likely be changes in climate and minor mass extinctions.
The “holy grail” of their supernova work is matching a detected one with a dramatic event in the Earth biosphere, most especially a mass extinction. The 2 to 3 million years ago period includes the boundary between the Pleistocene and Pliocene epochs, when Earth climate changed and major glaciations periods began — possibly supernova-related changes but not the extreme change a close supernova could produce.
Another potential effect of the supernova event of 2 to 3 million years ago is increased rates of mutation and of lightning, and thus forest fires on Earth.
Adrian Melott of the University of Kansas suggested that expected mutations from radiation sources such as supernovae could explain evolutionary changes in a variety of groups of organisms and creatures during that period — as a result of increased deadly cancers in some species and increased positive mutations in others.
He also said that evidence of more widespread wildfires during that long period — as measured in charcoal deposits — could be the result of increased cloud to ground lightning induced by the additional high-energy particle environment created by a relatively close supernova explosion.

The iron-60 signatures of a close supernova have been a great boon to the field, but they do not go back beyond that almost 10 million year period when the radioactivity was present. To go back further than that, Fields said different radioactive signatures would be needed — and not those that go back to the formation of the planet.
“It’s a hard problem because nature has been unkind,” he said. “The early mass extinctions – 100 million and more years ago – need radioactivity that lasts that long. And the only element we’ve found is plutonium-244, which is not stable in any form.”
Plutonium-244 has a half life of 80 million years, and so could potentially be used to identify close supernova explosions in a manner similar to iron-60, but during that much longer time frame. And as Fields explained it, plutonium-244 is produced in a few dramatic ways: during the explosion of a nuclear bomb, the explosion of a supernova, or the merging of a pair of neutron stars.”
Although the science around the formation and detection of plutonium-244 in nature is immature, he said it remains the best pathway to find that “holy grail” — a known mass extinction directly associated with a close supernova explosion.

